Organisation
The Chemical Stress Ecology Group is centered around the appointment of Paul Van den Brink at the Aquatic Ecology and Water Quality Management group of Wageningen University. The group members consists of PhD and MSc students and people of Wageningen University and Wageningen Environmental Research who work in close cooperation with Paul.
Scientific Interests
Some people working in the fields of ecotoxicology and ecological risk assessment aim to transform it into chemical stress ecology by incorporate more ecological theory and to achieve this by introducing novel methods, among others, traits based approaches and mechanistic effect models (Van Straalen, 2003; Van den Brink, 2008; 2013; Van den Brink et al., 2013). This to improve the ecological risk assessment of chemicals but also to obtain a better linkage with the assessments of effects of emerging chemicals and multiple stressors, the latter also including non-chemical stressors like climate change, habitat destruction and flow alteration (Moe et al., 2013; Van den Brink et al., 2016). This aim is, and will be in the future, one of the central passions of our work.
We believe that by focussing on developing new tools that facilitate a better integration of ecology into chemical stress ecology on the one hand and studying the effects of novel (combination of) stressors on the other hand, a good combination of fundamental science which can be practically used in the future is obtained.
a) New tools: traits-based and ecological modelling approaches
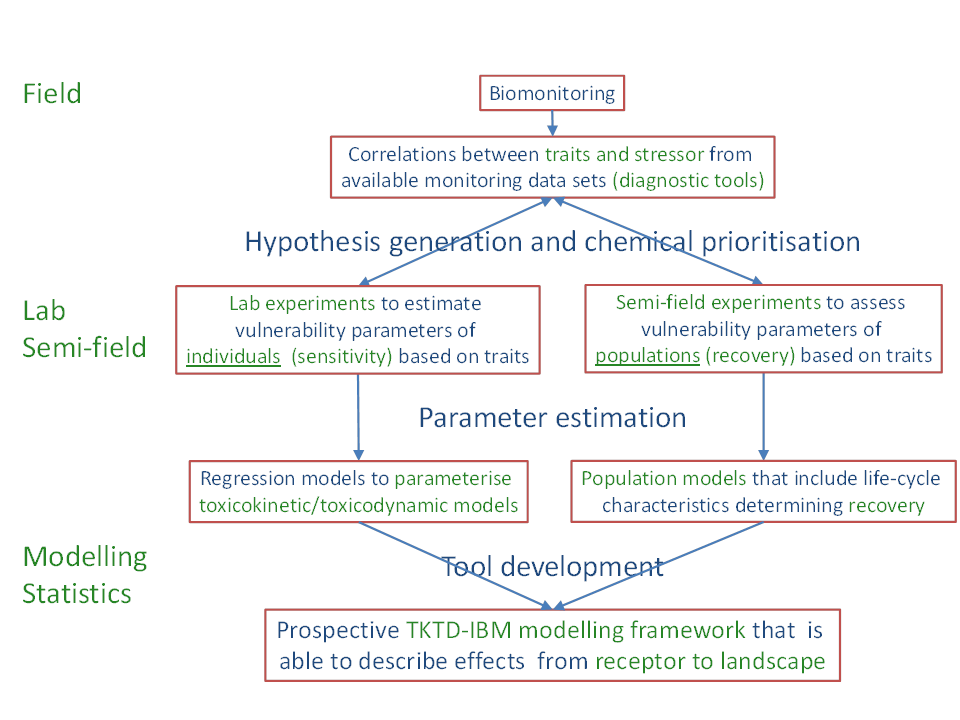
The tools described below all contribute to the development of a prospective modelling framework that is able to describe and predict the effects of chemicals at the individual, population and ecosystem level. This framework will use traits instead of taxonomy since they have a mechanistic relationship with the vulnerability of ecological components (response traits) and the ecological consequences of them being affected (effect traits). These mechanistic relationships will be captured in toxicokinetic-toxicodynamic models as well as individual based models and food-web models to describe the effects of chemicals at the individual, population and ecosystem level, respectively. Figure 1 shows the conceptual framework that incorporates retrospective traits-based approaches and prospective ecological models.
One of the holy grails of chemical stress ecology is to find out which species characteristics (traits) make one species more sensitive to a chemical stressor than another one (Baird and Van den Brink, 2007). In the past, two approaches have been used to assess the (cor)relationships between species traits and their sensitivity, one based on empirical correlations (Rubach et al., 2010a) and one based on a more mechanistic approach using toxicokinetic/toxicodynamic experiments and models (Rubach et al., 2012). In the latter paper it is shown that almost 90% of the variation in uptake and 80% of the variation in elimination of an insecticide in a range of 15 freshwater arthropod species can be explained by 4 species traits. These traits were surface area (without gills), detritivore, using atmospheric oxygen and phylogeny in case of uptake and thickness of exoskeleton, complete sclerotization, using dissolved oxygen and % lipid of dry weight in case of elimination. For most of these traits a mechanistic hypothesis between the traits and their influence on the uptake and elimination can be made (Rubach et al., 2011; 2012). Rubach et al. (2012) also showed that a large amount (50–60 %) of the variation in sensitivity (48h,EC50) can be explained by the toxicokinetics, i.e. by uptake (32 %) and elimination (28 %) (Rubach et al. 2010b), while the remaining variation (40–50 %) might therefore be attributed to biotransformation, distribution of the oxon or the toxicodynamics. This shows that it is possible to construct mechanistic models that are able to predict the toxicokinetics of chemicals in species based on their traits, although this has only been studied for one chemical at the moment and no systematic work has been done yet on the toxicodynamic side (this research constitutes the left column of Figure 1).
We would like to follow up this mechanistic work on the traits-sensitivity (cor)relations. Since this research is able to answer some quite fundamental questions of ecotoxicology.
Figure 1. Conceptual framework that incorporates retrospective traits‐based approaches and prospective ecological models that is able to describe the effects of chemicals from the receptor to the landscape level. This framework incorporates data from field monitoring studies, laboratory toxicity studies, and semi-field experiments (adapted from Van den Brink et al., 2013).
We have quite some PhD projects that involve chemical and biological monitoring, i.e. the projects of Fengjiao Peng, Kizar Ahmed Sumon, Michael Onwona-Kwakye, Concillia Monde, Zhang Naisheng, Berhan Teklu and Jadipa Khatikarn. One of the problems of field monitoring is that the effects of single stressors have to be separated from many others, which are often even correlated with the stressor of interest. To overcome this diagnostic problem in monitoring, traits-based approaches have been proposed (Figure 1 and Van den Brink et al., 2011). The singular failure of traits-based approaches to ‘penetrate the biomonitoring market’ yet, however, shows that scientists in this area need to raise their game in terms of relevance and closer attention to the needs of ERA practitioners, who generally seek insight into causal mechanisms rather than subjective narratives based on phenomenology. Some studies, however, already illustrate the potential of trait patterns observed at community level to yield specific diagnostic information to an individual stressor, based simply on the observation of taxon occurrence in relation to specific habitat variables (top row of Figure 1, e.g. Horrigan and Baird, 2008; Statzner and Bêche, 2010). The above listed PhD projects and the EU SOLUTIONS project will be used to further develop these traits-based monitoring tools to address the multiple stress issue under field situations (Brack et al., 2015; Van den Brink et al., 2011; 2013; Altenburger et al., 2015). In order to use these tools and their results for environmental diagnosis it is pivotal that ecological scenarios are developed which should include biotic parameters (living organisms) and their associated ecological functions and/or interactions and abiotic parameters that influence the chemical exposure, their toxic effects and/or the recovery process (Rico et al., 2016).
In order to bring the science of chemical stress ecology beyond the level of phenomenology, it is important that the experience gathered in field monitoring and laboratory and semi-field experiments on the direct and indirect effects of chemicals and the recovery of populations is captured in ecological models (Figure 1, Van den Brink, 2013; Van den Brink et al., 2013). For simplicity, we can distinguish between ecological models at three levels of biological organisation, the individual, the population and the ecosystem level. In our case we use toxicokinetic/toxicodynamic models, individual based models and food-web models to describe the effects at these three levels, respectively (e.g. Galic et al., 2014; Baveco et al., 2014; Traas et al., 2004). The use of ecological models in the risk assessment of chemicals was highly facilitated by the organisation of the LEMTOX workshop (Forbes et al., 2009), the finished CREAM (Grimm et al., 2009) and EPOCH projects (Galic et al., 2010; Hommen et al. 2010) and the ongoing ChimERA (De Laender et al., 2014) and Solutions (Brack et al., 2015; Altenburger et al., 2015.) projects. While the CREAM and EPOCH projects aimed to develop individual based models to assess the effects on and recovery of populations (e.g. Focks et al., 2014), the ChimERA and Solutions projects aim to develop multiple species (food-web) models and to address the problem of mixture and multiple stress using ecological models.
Using these and future projects, we would like to dive more into the ecological modelling and their use for risk assessment to support the ecosystem and landscape level risk assessment of multiple stressors, including non-chemical ones. It is hypothesised that the use of single species individual-based models yield invalid predictions since species interactions like competition and predation are not explicitly included. Within the ChimERA project we have performed experiments on these interactions (Del Arco Ochoa et al., 2015a;b) and develop multi-species models in order to test this hypothesis (De Laender et al., 2015; Viaene et al., acc.) and reality showed that chemical effects are sometimes, but not always, enhanced by ecological interactions. The further development of landscape level ecological models is greatly needed to assess the effects of continuous exposure to a single stressor in a spatially explicit landscape (Focks et al., 2014) and the effects of multiple stressors which act at different spatio-temporal scales (Van den Brink et al., 2016). These issues will be addressed in the PhD project of Sanne Van den Berg which will focus on the development of ecological scenarios and models for use in the risk assessment of HPCs.
b) Emerging chemicals and multiple stressors
Although toxic effects on aquatic life are regularly observed, it remains a great challenge to link occurrence of chemicals with the ecological status of waters, to identify major chemical stressors that would call for management, and to find efficient solutions for the abatement of pollution-related risks (Altenburger et al., 2015; Brack et al., 2015). Typically, complex mixtures of priority pollutants, emerging substances, by- and transformation products, and natural compounds co-occur in aquatic systems, thus rendering a comprehensive assessment challenging. Emerging substances include a multitude of polar and even ionic compounds for instance belonging to the groups of veterinary medicines and/or household and personal care products (HPCs) and for which many of the tools and models developed for classical non-polar persistent organic pollutants (POPs) do not apply (Brack et al., 2015; Altenburger et al., 2015). For instance, Rico et al. (2014a) and Andrieu et al. (2015) showed that antibiotics, disinfectants and parasitics may pose ecological risks for aquatic ecosystems neighbouring Asian aquaculture (especially Pangasius catfish production in Vietnam), although data availability is scarce and herewith the uncertainty of the risk assessment also becomes large. Rico et al. (2014b) studied the use, environmental fate and ecological risks of antibiotics applied in tilapia cage farming in the Tha Chin and Mun rivers in Thailand. Their results indicate insignificant short-term risks for primary producers and invertebrates, but suggest that the studied aquaculture farms constitute an important source of antibiotic pollution with unknown long-term consequences for the aquatic ecosystem, which may be expected for especially the sediment compartment. Zhang et al. (subm.) measured some HPCs (biocides, benzotriazoles and UV filters) chemicals in surface water and sediment of the Sha Rivers, South China an concluded that triclosan and clotrimazole exceeded safe threshold values in the water phase while triclocarban, carbendazim and climbazole posed high risk in the sediment compartment and methylparaben, triclocarban benzotriazole and 5,6-dimethylbenzotriazole posing median risks to sediment associated aquatic organisms. These studies show that on the one hand chemical monitoring data are missing for emerging chemicals while on the other hand data on their fate and effects are also lacking (Brack et al., 2015; Rico et al., 2012). In the ongoing Solutions project and PhD projects of Fengjiao Peng, Zhang Naisheng and Jadipa Khatikarn we aim to address these issues.
Aquatic sediments are an important part of the aquatic ecosystem, providing critical ecosystem processes and services. The sediment compartment acts as a sink for hydrophobic organic chemicals, which might affect the processes and services provided and, herewith, become emerging chemicals although there presence is already known for quite some time. Therefore, sediment should be considered in environmental risk assessment of emerging chemicals but is currently underrepresented. Current sediment research needs are the harmonization of test protocols and the development of approaches to translate biodegradation process parameters obtained from lab tests to parameters that are relevant for the field situation (Diepens et al. 2014). During the PhD project of Noel Diepens, we got involved in assessing the toxicity of sediments. She reviewed the available literature on whole sediment toxicity tests for microorganisms, benthic invertebrates, macrophytes and benthic communities (Diepens et al., 2014), performed experiments to evaluate routes of chemical uptake for invertebrates and macrophytes and the effects of chemicals on sediment associated bacteria and developed a model for a benthic invertebrate (Chironimus riparius) (Diepens et al., 2016). Diepens et al. (2017) states that the combination of experimental work, fate modelling and effect modelling allows for clear perspectives of better and more cost-effective prognostic tools for sediment risk assessment. With the PhD projects of Fengjiao Peng, Zhang Naisheng and Jadipa Khatikarn in South-East Asia, the toxicity of polluted sediments becomes a central theme since these projects focus on household and personal care chemicals (HPCs) that are continuously emitted and bound to the organic fraction in the sediment. HPCs exhibit a wide range of physical-chemical properties which influence their fate and effects in the environment. Especially the hydrophobic HPCs pose particular challenges for aquatic risk assessments. Compounds with a strong binding affinity for living or non-living organic particles will sorb to these particles in the water column and subsequently concentrate in sediments, often to levels several orders of magnitude higher than those found in surface waters. The persistence, fate and effects of sediment-associated compounds depend on a variety of factors including their bioavailability to bottom-living organisms and their subsequent susceptibility to biotransformation. In the sediment related project we aim to determine the effects of chemical on sediment associated aquatic populations as well as the effects of these populations on the fate of these chemicals as especially deposit feeders may be important in facilitating the removal of contaminants from the sediment.
In 1999, a SETAC workshop was held in Pellston, Michigan, focusing on the integration of the best-available ecological science within the field of ecotoxicology (Baird and Burton, 2001). In particular, the value of ecological models was highlighted as a potentially fruitful area for cross-fertilisation between the two disciplines, as was the need for better tools for environmental management. In 2014, the world is a very different place, as impacts of rapid global change processes are now an almost routine topic of conversation in the news media. Yet, despite a considerable body of published research, answering the question: “how can we manage threatened ecosystems effectively?” remains poorly supported by traditional tools and approaches. Faced with multiple threats to natural ecosystems, each with their own unique signature effects and consequences, our regulatory systems remain over-reliant on single-substance guidelines, and fail to deal effectively with large-scale environmental change processes either as drivers in their own right, or as modifiers of local anthropogenic stressor regimes. A recent attempt to synthesise the effects of large-scale drivers as modifiers of local stress regimes, and the resulting effects on ecosystem structure and function (Vörösmarty et al, 2010) clearly illustrates the value of attempting to capture the complexity of pattern and process changes at a global scale, but also highlights the limitations of such approaches where local-scale information on key drivers, particularly in terms of toxic substance emissions remain difficult to access. With recent advances in key areas such as ecological modelling, biomonitoring science, ecogenomics and earth observation technologies – many of which are truly ground-breaking – significant new opportunities for the study of complex stress regimes are now available (e.g. Baird and Hajibabaei, 2013; Van den Brink et al, 2013; 2016). The activities described above will aim:
- to explore the potential offered by these new approaches to characterise stressor regimes,
- to explore stressor-response relationships among biota also using Big Data,
- to design better early-warning systems using traits based and genetic tools,
- to develop mechanistic, ecological response models and
- to develop ways to link multiple chemical occurrence to the assessment of biological adverse effects of mixtures.
By doing so smart tools are developed to support sustainable management of human activities through more efficient regulation. This because future RAs will be probabilistic, spatial explicit, ecological and will include effects on ecosystem services (e.g. Galic et al., 2012). These risk assessments will be part of adaptive management and will be explicit about the trade-offs (social values vs environmental consequences) and will be connected with human health and well-being. The current and future work conducted in several PhD (Fengjiao Peng, Zhang Naisheng and Jadipa Khatikarn) and other (Solutions and ChimERA) projects as well as the experience gathered during a recent workshop on multiple stressors (Van den Brink et al., subm.) that Paul recently co-organised will be brought together in this recent theme. The genetic tools and experience they provide is essential for studying the effects of stressors on the structure and functioning of microbial assemblages as well as to develop early-warning systems based on gene expression (Dimitrov et al., 2014; Rico et al., 2014; Diepens et al., 2014).
References not provided in the “publication” section:
Baird, D.J. and G.A. Burton (eds.). 2001. Ecological Variability: Separating Anthropogenic from Natural Causes of Ecosystem Impairment. SETAC Press, Pensacola, USA.
Baird, D.J. and M. Hajibabaei. 2012. Biomonitoring 2.0: a new paradigm in ecosystem assessment made possible by next-generation DNA sequencing. Molecular Ecology, 21, 2039-2044.
Horrigan, N., and D.J. Baird. 2008. Trait patterns of aquatic insects across gradients of flow‐related factors: A multivariate analysis of Canadian national data. Can J Fish Aquat Sci 65:670–680.
Rubach, M.N., R. Ashauer, D.B. Buchwalter, H.J. De Lange, M. Hamer, T.G. Preuss, K. Töpke and S.J. Maund. 2011. A framework for traits‐based assessment in ecotoxicology. Integr Environ Assess Manag 7:172–186.
Statzner B, Bêche LA. 2010. Can biological invertebrate traits resolve effects of multiple stressors on running water ecosystems? Freshwater Biol 55:80–119.
Van Straalen, N. M. Ecotoxicology becomes stress ecology. Environ. Sci. Technol. 2003, 37, 324A–330A
Vörösmarty, C.J., P.B. McIntyre, M.O. Gessner, D. Dudgeon, A. Prusevich, P. Green, S. Glidden, S.E. BunnE, C.A Sullivan, C. Reidy Liermann and P.M. Davies. 2010. Global threats to human water security and river biodiversity. Nature, 467, 555–561.
Facilities
Link to the indoor laboratories of the Environmental Risk Assessment Group of Wageningen Environmental Research
Link to the outdoor cosm facilities of the Environmental Risk Assessment Group of Wageningen Environmental Research